General Information
- Chlorotoxin is a small positively charged toxin from the Deathstalker scorpion.
- This toxin is a 36 amino acid peptide, with 8 cysteines that form 4 disulfide bonds within it.
- Consists of a small three-stranded antiparallel beta-sheet packed against an a-helix, thereby adopting the same fold as charybdotoxin but chlorotoxin has an extra disulfide bridge that charybdotoxin lacks, this extra bridge changes the electrostatic surface charge making it very positive. (1)
Mechanism of Action
 http://myscienceacademy.org/2013/07/09/worlds-deadliest-scorpion/
http://myscienceacademy.org/2013/07/09/worlds-deadliest-scorpion/
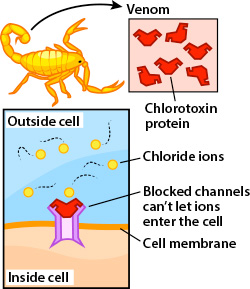
- Chlorotoxin works first by being injected into the body through the stinger of the scorpion.
- Once in it targets the small conductance chloride channels and blocks the channel. (2)
- Each Chlorotoxin can only bind to one chloride channel so the more channels available the more of the toxin is needed. (3)
- Binding to the chloride channels in muscles leads to complete paralysis of the organism due to a lack of the cell membrane to reach its resting potential.
- Paralysis is reversible since the Chlorotoxin is a competitive protein, not permanent.
By: Steven Melter
- Lippens, G., Najib, J., Wodak, S. J., & Tartar, A. (January 01, 1995). NMR sequential assignments and solution structure of chlorotoxin, a small scorpion toxin that blocks chloride channels. Biochemistry, 34, 1, 13-21.
- Crow, James Mitchell. "Drugs With Bite." New Scientist 214.2863 (2012): 34-37. Computers & Applied Sciences Complete. Web. 27 Mar. 2015.
- J.A. DeBin, G.R. Strichartz, Chloride channel inhibition by the venom of the scorpion Leiurus quinquestriatus, Toxicon, Volume 29, Issue 11, 1991, Pages 1403-1408, ISSN 0041-0101, http://dx.doi.org/10.1016/0041- 0101(91)90128- E. (http://www.sciencedirect.com/science/article/pii/004101019190128E)
